Hantavirus Outbreak Raises Questions About Isolation Capacity and Healthcare Surge Preparedness
Recent international reports involving a suspected outbreak of the Andes strain of hantavirus have prompted the U.S. Centers for Disease Control and Prevention (CDC) to activate a Level 3 emergency response — the agency’s lowest emergency activation level, but one that still signals heightened monitoring, coordination, and preparedness efforts.
While public health officials continue to emphasize that the overall risk to the general public remains low, the outbreak has renewed discussions about healthcare surge readiness, negative-pressure isolation capacity, and rapidly deployable infectious disease treatment infrastructure.
For healthcare systems, emergency planners, and government agencies, the situation serves as another reminder that hospitals and treatment facilities must remain prepared for infectious disease events capable of overwhelming existing isolation resources.
What Is Hantavirus?
Hantaviruses are a family of viruses primarily carried by rodents. Human infection typically occurs through exposure to aerosolized particles from infected rodent urine, droppings, or saliva.
In the Americas, hantavirus infections can lead to Hantavirus Pulmonary Syndrome (HPS), a severe respiratory illness that may rapidly progress to respiratory failure and death. Fatality rates for severe cases have historically approached 35%–40%.
Early symptoms often resemble influenza or other viral respiratory illnesses, including:
- Fever
- Fatigue
- Muscle aches
- Headache
- Chills
- Nausea
- Abdominal discomfort
As the illness progresses, patients may develop:
- Severe shortness of breath
- Pulmonary edema
- Respiratory distress
- Low oxygen levels
- Cardiovascular instability
Because early symptoms can appear relatively nonspecific, infected individuals may continue traveling, interacting with others, or seeking routine medical care before the disease is recognized.
Why the Andes Variant Is Different
Most hantavirus strains are not believed to spread from person to person. However, the Andes virus (ANDV) — primarily found in Argentina and Chile — is the only known hantavirus strain with documented human-to-human transmission.
Health authorities investigating the recent cruise ship outbreak have indicated that laboratory testing strongly suggests the Andes strain may be involved.
According to the World Health Organization (WHO), transmission between people appears uncommon and generally requires close, prolonged contact, often involving household members or intimate partners.
It’s important to note that WHO and CDC officials have stated there is currently no evidence that asymptomatic individuals significantly spread the virus.
Still, the possibility of limited human-to-human transmission changes how healthcare systems approach isolation protocols, patient triage, and infection control procedures.
Incubation Period and Travel Concerns
One of the most concerning aspects of hantavirus infection is its potentially long incubation period.
Current health guidance indicates symptoms may develop anywhere from 1 to 8 weeks after exposure, though many cases appear within 2 to 4 weeks.
This prolonged incubation window creates operational challenges for public health agencies because exposed individuals may:
- Travel internationally or domestically before symptoms develop
- Interact with family members, coworkers, and healthcare personnel
- Board aircraft, cruise ships, or public transportation
- Require extensive contact tracing once diagnosed
Recent reports indicate that passengers potentially exposed during the cruise ship incident have already returned to multiple countries, including the United States.
Although officials continue to stress that the risk of widespread transmission remains low, the outbreak highlights how modern travel can rapidly complicate efforts to contain infectious diseases.
What a CDC “Level 3” Emergency Response Means
To help coordinate federal public health responses during emerging events, the CDC uses a tiered activation structure: Level 1, Level 2, and Level 3 — each reflecting the scale, complexity, staffing requirements, and operational response needed to address a public health emergency. The activation level helps determine how resources, personnel, coordination efforts, and emergency operations are deployed across local, national, and international response agencies. The information below provides a more in-depth explanation of each CDC emergency activation and response level.
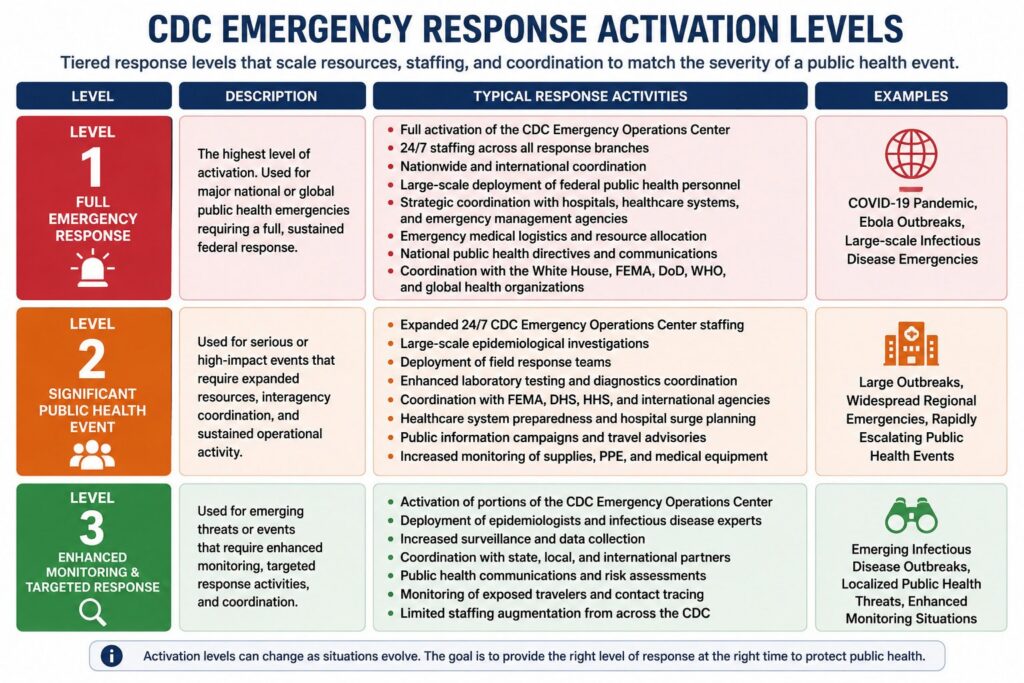
Level 1 Activation — Full Emergency Response
A Level 1 activation is the CDC’s highest emergency response level and is reserved for major national or global public health emergencies.
During a Level 1 response, the CDC operates at maximum emergency capacity with agency-wide support and sustained around-the-clock operations.
A Level 1 activation may include:
- Full activation of the CDC Emergency Operations Center
- 24/7 staffing across multiple response branches
- Nationwide and international coordination efforts
- Large-scale deployment of federal public health personnel
- Strategic coordination with hospitals, healthcare systems, and emergency management agencies
- Emergency medical logistics and resource allocation
- Expanded quarantine and isolation guidance
- National public health directives and operational support
- Coordination with the White House, FEMA, DoD, WHO, and global health organizations
Examples of events that have involved Level 1 activation include the COVID-19 pandemic, major Ebola responses, and other large-scale infectious disease emergencies with significant international impact.
Level 2 Activation — Significant Public Health Event
A Level 2 activation represents a more substantial and resource-intensive response. These events generally require broad interagency coordination, expanded staffing, and sustained operational activity.
A Level 2 response may involve:
- Expanded 24/7 CDC Emergency Operations Center staffing
- Large-scale epidemiological investigations
- Increased deployment of field response teams
- Enhanced laboratory testing and diagnostic coordination
- Coordination with FEMA, DHS, HHS, and international agencies
- Healthcare system preparedness guidance and hospital surge planning
- Expanded public information campaigns and travel advisories
- Increased monitoring of supply chains, PPE, and medical equipment
Level 2 activations are commonly associated with large infectious disease outbreaks, widespread regional emergencies, or rapidly escalating public health situations that could strain healthcare systems.
Level 3 Activation — Enhanced Monitoring and Targeted Response
The current hantavirus response has reportedly been classified as a Level 3 activation — the lowest formal emergency response level. Even at this level, the CDC initiates structured coordination and heightened monitoring activities.
A Level 3 response may include:
- Activation of portions of the CDC Emergency Operations Center
- Deployment of specialized epidemiologists and infectious disease experts
- Increased surveillance and data collection
- Coordination with state, local, and international health agencies
- Public health communications and risk assessments
- Monitoring of exposed travelers and contact tracing operations
- Limited staffing augmentation from across the CDC
Level 3 activations are typically used for emerging infectious disease outbreaks, localized public health threats, or situations requiring enhanced federal coordination without overwhelming the national healthcare infrastructure.
Why the Activation Level Matters
While a Level 3 activation does not indicate a pandemic declaration or imminent widespread threat, it signals that federal health authorities consider the situation serious enough to require organized national monitoring and response coordination.
For hospitals, healthcare systems, emergency planners, and medical infrastructure providers, even lower-level activations can serve as an early warning to evaluate:
- Isolation room capacity
- Negative pressure treatment capabilities
- Surge hospital readiness
- Infectious disease triage protocols
- Availability of rapidly deployable medical infrastructure
- Staffing and PPE preparedness
As seen during COVID-19, public health emergencies can escalate rapidly. Early preparedness planning often determines how effectively healthcare systems can respond if conditions worsen.
The Importance of Negative Pressure Isolation Capacity
Outbreaks involving respiratory illnesses with potential person-to-person transmission place immediate pressure on healthcare systems to isolate infectious patients safely.
Negative-pressure isolation environments are designed to help prevent contaminated air from escaping from patient treatment areas by maintaining a lower air pressure inside the room than in adjacent spaces. These systems are commonly used for:
- Airborne infectious disease treatment
- Isolation wards
- Emergency surge hospitals
- Quarantine operations
- Infectious disease triage centers
- Biocontainment operations
During the COVID-19 pandemic, healthcare systems worldwide experienced significant shortages of isolation rooms and surge treatment space. Similar challenges could recur if future infectious disease events require rapid expansion of patient isolation capacity.
Rapidly Deployable Isolation and Treatment Infrastructure
Healthcare preparedness increasingly depends on the ability to rapidly expand treatment capacity without waiting months or years for permanent construction projects.
BLU-MED Response Systems® develops rapidly deployable medical infrastructure solutions designed to support healthcare surge operations, disaster response, infectious disease treatment, and emergency medical expansion requirements.
The BLU-MED® Negative Pressure Isolation Shelter System provides rapidly deployable negative pressure treatment and isolation capabilities that can be configured to support:
- Temporary infectious disease isolation wards
- Surge hospital expansion
- Quarantine facilities
- Emergency triage operations
- Military medical operations
- Humanitarian response missions
- Remote or austere healthcare environments
These rapidly deployable systems can help healthcare organizations and emergency response agencies increase isolation capacity quickly when traditional hospital infrastructure becomes strained.
Preparedness Before Crisis
At this stage, health officials continue to emphasize that the risk of widespread hantavirus transmission remains low.
However, recent global outbreaks — from COVID-19 to Ebola, SARS, and emerging influenza variants — have repeatedly demonstrated the importance of proactive healthcare preparedness.
The current hantavirus situation underscores several ongoing realities:
- Infectious disease outbreaks can emerge rapidly
- International travel accelerates exposure pathways
- Isolation capacity remains a critical healthcare vulnerability
- Healthcare surge infrastructure must be rapidly scalable
- Preparedness planning is far easier before hospitals become overwhelmed
As public health agencies continue monitoring the outbreak, healthcare providers, emergency planners, and government organizations may once again evaluate whether existing isolation and treatment infrastructure is sufficient to respond to future infectious disease events.