Antibody Infusion Centers for COVID-19
One of the newest strategies in the fight against COVID-19 is antibody infusion therapy. Here’s what you need to know about antibodies, their effectiveness against the novel coronavirus, and setting up safe antibody infusion centers.
What Are Antibodies?
Antibodies are proteins that our bodies naturally create to prevent viruses from spreading. When antibodies spot viruses enter your body, they bind to them to block them from entering your cells. This blockage is crucial for preventing diseases from developing in the first place, and from progressing any further.
Convalescent Plasma Therapy
Those who recover from a specific virus have antibodies in their blood that may be effective in preventing others from being infected or diminishing the negative effects of the virus. That’s where convalescent plasma therapy comes in.
Convalescent plasma therapy takes blood from recovered patients and processes it to remove the blood cells, leaving behind just the liquid plasma and antibodies. The plasma and antibodies are then transfused into sick patients to reduce the severity or length of the disease and increase their ability to fight it.
Blood plasma therapy has been proven effective at treating diseases such as the Spanish flu of 1918 as well as the more recent SARS and Ebola outbreaks.
Monoclonal Antibody Therapy
If an antibody taken through convalescent plasma therapy is discovered to be effective in treating or preventing a disease, it can be duplicated in a lab for widespread use.
The resulting lab-made proteins are called monoclonal antibodies. By mimicking the immune system’s ability to fight off viruses, monoclonal antibodies can help prevent and treat diseases just like the antibodies produced naturally in our bodies.
Monoclonal antibody therapy, or the transfusion of lab-made antibodies into patients, has been used to treat certain types of cancer and is currently being reviewed by the FDA as a means for treating Ebola.
Because lab-made antibodies may not last as long as those naturally produced, multiple infusions are sometimes necessary for prolonged effectiveness.
[Related: Airborne Diseases: An Overview]
How Antibody Infusions Are Being Used to Fight COVID-19
The fact that antibody infusions have been successful in treating SARS (a respiratory illness caused by another type of coronavirus) has led experts to think it may be effective in treating COVID-19, and this has turned out to be the case.
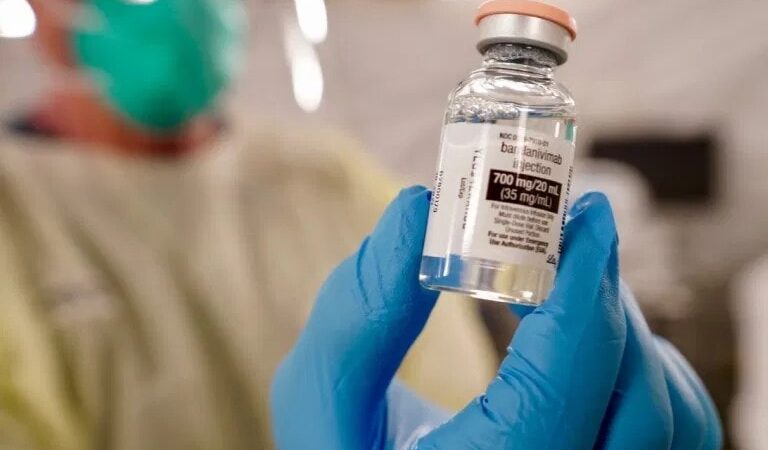
In November of 2020, the U.S. Food and Drug Administration (FDA) issued emergency-use authorizations (EUAs) for two monoclonal antibody drugs for the treatment of COVID-19, bamlanivimab and a combination of casirivimab and imdevimab. Both of these therapies were shown in clinical trials to reduce hospitalization and emergency room visits in high-risk patients within the first 28 days post-treatment.
The goal of these monoclonal antibodies, which are the only drugs approved for use prior to hospitalization, is to prevent additional strains on the already overtaxed hospital infrastructures across the country. By administering the therapies to newly diagnosed high-risk patients, healthcare facilities hope to keep the disease from progressing and prevent future hospitalizations.
With the COVID-19 vaccine rollout taking longer than expected, and herd immunity expected fall of 2021 at the earliest, preventing hospital overcrowding will be essential to the continued fight against the coronavirus. And monoclonal antibodies may just be the solution.
[Related: Key Elements of Pandemic-Ready Healthcare Facilities]
How BLU-MED Medical Shelters Help Establish Safe Antibody Infusion Centers

Antibody infusion centers have already been set up in various locations across the U.S., primarily in areas where hospitals and/or intensive care units (ICUs) have sustained a 0% capacity for a prolonged period.
Just like with COVID testing and vaccination centers, the challenge in setting up antibody infusion centers is the risk of doing more harm than good. That is, by establishing an antibody infusion center within existing healthcare facilities, you may expose staff and patients to the virus and promote transmission.
For that reason, it’s preferred to create a separate, isolated facility designated solely for antibody infusions.
Medical shelters from BLU-MED Response Systems® are ideal for creating these facilities: Rapidly deployable, ruggedly durable, and highly customizable, BLU-MED shelters can help quickly establish antibody infusion centers of any size, with just about any requirements, in any climate.
Because BLU-MED medical shelters include options for insulation as well as HVAC units, they are able to provide a comfortable environment for patients and healthcare staff to wait out the two and a half hours it takes to administer the infusion and monitor for adverse reactions.
In fact, several healthcare facilities have already proven the effectiveness of using BLU-MED medical shelters to set up antibody infusion centers:
- Mountain Area Health Education Center (MAHEC) in Asheville, North Carolina
- Covenant Health at Covenant Medical Center in Lubbock, Texas
- Imperial County Infusion Center at El Centro Regional Medical Center (ECRMC) in El Centro, California
Covenant Health is hoping their 14-bed BLU-MED medical shelter will help reduce their 139 hospitalized COVID patients by up to 26 patients, and the Imperial County Infusion Center’s eight-bed shelter is expected to treat 28 to 30 patients a day, seven days a week.
For a closer look at Covenant Health’s antibody infusion center, watch the video below. For more information on BLU-MED medical shelters and how they can help you establish an antibody infusion center of your own, reach out to one of our building specialists today.
Featured image courtesy of the Department of Human and Health Services